
Shelagh Fogarty 1pm - 4pm
31 December 2020, 18:33 | Updated: 31 December 2020, 18:49

China has recorded its first case of the new highly-contagious coronavirus variant that was first seen in the UK.
The new strain was detected in a 23-year-old female student who had returned to the Far East nation from Britain, according to the Chinese Center for Disease Control and Prevention (CDC).
She was tested for coronavirus in Shanghai on 14 December after showing mild symptoms and she was hospitalised upon arrival, the latest edition of China CDC weekly reported.
Health experts tested her samples on 24 December “due to travel history from the UK and abnormalities in nucleic acid test results”, the China CDC said.
The publication added that the infection "poses a great potential threat to the prevention and control of Covid-19 in China".
Britain's variant has prompted more than 50 countries to set up travel restrictions on the UK, including where Covid originated - China.
Read more: China approves state-owned coronavirus vaccine
Read more: Millions to get first dose of Oxford vaccine sooner

Luke from China - Update 11 months on
It comes on New Year's Eve, marking exactly one year since the World Health Organisation first mentioned a mysterious pneumonia-like disease in China, which was later identified as Covid-19.
The virus has gone on to kill nearly 2 million people and has brought markets and the global economy to its knees.
Earlier on Thursday, China granted “conditional” market approval to its Sinopharm vaccine which has a reported 79 per cent efficacy rate, Chinese health authorities said.
Roughly 4.5 million shots of the jab have already been administered to health workers and others who work in overseas jobs.
Neighbouring Pakistan has plans to purchase 1.2 million doses of the Sinopharm vaccine, the Islamic country's minister for science and technology said on Twitter.
Read more: China's lunar capsule brings home rocks from moon
Read more: Australia demands apology from China over fake post of soldier murdering child
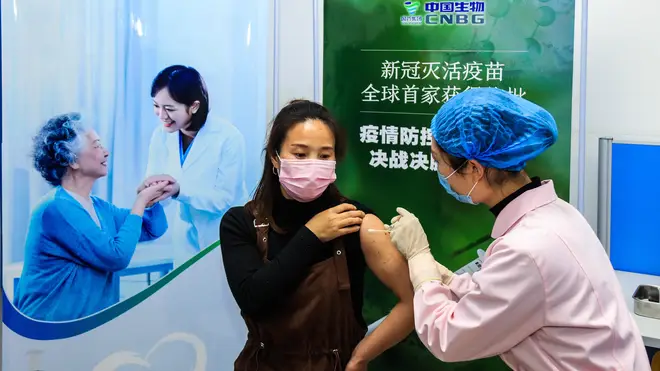

The two-dose Covid drug is the first approved for general use in the Far East nation and its approval comes as the country begins vaccinations for 50 million people ahead of the Lunar New Year holiday in February.
Chen Shifei, the deputy commissioner of China's National Medical Products Administration, told a news conference that conditional approval means research is still ongoing and the company will be required to submit follow-up data, as well as reports of any adverse effects after the jab is sold on the market.
The vaccine was developed by the Beijing Institute of Biological Products, a subsidiary of state-owned conglomerate Sinopharm.
It is an inactive vaccine, which means the virus was grown in a lab and then killed. The germ is then injected into the body to generate an immune response. Final proof of its effectiveness will depend on publication of more data.
The drug has been approved in the United Arab Emirates and Bahrain, and is slated for use next in Morocco.
Listen & subscribe: Global Player | Apple Podcasts | Google Podcasts | Spotify