
Nick Abbot 10pm - 1am
4 February 2021, 13:59 | Updated: 4 February 2021, 14:03
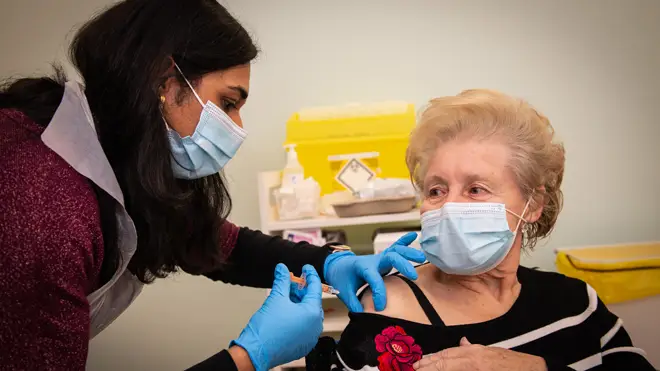
Vaccines minister Nadhim Zahawi has told MP's that the UK is "getting safer every day" because fo the rollout of coronavirus jabs.
Mr Zahawi told MPs that the government is on track to protect the four most vulnerable groups by mid-February, aiming to reach a target of 15 million first jabs by February 15.
Mr Zahawi hailed the success of the UK's vaccination programme, telling the Commons: "Our nation is getting safer every day."
"I am pleased to inform the House that we have now vaccinated almost nine in 10 over-80s in the UK, almost nine in 10 over-75s, and over half of people in their 70s," he said.
"We've also visited every eligible care home possible with older residents in England and have offered vaccinations to all their residents and staff.
Nadhim Zahawi: We WILL get to 15m jabs by mid-February target
"This means we are currently on track to meet our target of offering a vaccine to the four most vulnerable groups by mid-February."
Mr Zahawi spoke to LBC earlier today saying that he was confident that the mid-February target would be reached.
His remarks come after a new study was launched which aims to determine whether it is safe to use different coronavirus vaccines.
Jonathan Van Tam said he believed the strategy of mixing the vaccines could provoke an enhanced immune response.
The research, which has received £7 million in funding through the Government's Vaccines Taskforce, will determine whether a mixed-dose vaccine regimen is better than, or a good alternative to, using two doses of the same Covid-19 jab.
The study, dubbed Com-Cov, will initially look at mixing doses of the Oxford University/AstraZeneca and Pfizer/BioNTech vaccines, as well as different intervals between doses, but researchers said more vaccines will be added to the list as they get approved for use.
The team will also be looking to see whether mixing doses will offer protection against coronavirus variants.
Professor Van-Tam said: “Given the inevitable challenges of immunising large numbers of the population against Covid-19 and potential global supply constraints, there are definite advantages to having data that could support a more flexible immunisation programme, if needed and if approved by the medicines regulator.
“It is also even possible that by combining vaccines, the immune response could be enhanced giving even higher antibody levels that last longer; unless this is evaluated in a clinical trial we just won’t know.

“This study will give us greater insight into how we can use vaccines to stay on top of this nasty disease.”
If findings show the combined effect of the jabs is safe and induces a strong immune response, the researchers say it would make vaccine implementation more flexible and lay the groundwork for introducing additional booster doses, should they be needed.
Read more: PM says Covid-19 cases 'still alarmingly high' despite vaccine milestone
Read more: Professor Chris Whitty responds after being harangued by Covidiot in the street
A wait of 12 weeks between vaccine doses is best, Virologist says
Deputy chief medical officer and senior responsible officer for the study, Professor Jonathan Van-Tam, said: "Given the inevitable challenges of immunising large numbers of the population against Covid-19 and potential global supply constraints, there are definite advantages to having data that could support a more flexible immunisation programme, if needed and if approved by the medicines regulator.
"It is also even possible that by combining vaccines, the immune response could be enhanced giving even higher antibody levels that last longer; unless this is evaluated in a clinical trial we just won't know.
"This study will give us greater insight into how we can use vaccines to stay on top of this nasty disease."
Read more: 10 million people in UK have now received first dose of Covid vaccine
Read more: UK joins together in national clap for Captain Sir Tom Moore
The study will initially test eight different combinations, and include dosing schedules that are either 12 weeks apart or 28 days apart.
Those aged 50 and over are being called on to participate in the research, with 820 patients expected to take part.
They will be recruited over the course of February through the NHS Covid-19 Vaccine Research Registry, with initial results expected to become available during the summer - "in time to inform policy use of booster vaccines amongst younger cohorts".
Labour's Shadow Health Secretary praises vaccine rollout
Matthew Snape, associate professor in paediatrics and vaccinology at the University of Oxford and Chief investigator in the study, said: "We are looking to enrol adults aged 50 and over, allowing those who have mild-to-moderate comorbidities.
"And we're aiming to have recruitment from the black and minority ethnic communities that is representative of the UK population."
Prof Snape said those over 70s - who are at the greatest risk of severe Covid-19 disease - may have already been immunised which is why the recruitment age has been lowered to 50 and above.
The study is being run by the National Immunisation Schedule Evaluation Consortium (NISEC) across eight different sites across England - which includes London, Oxford, Southampton, Birmingham, Bristol, Nottingham and Liverpool.
The research is the first to determine the effects of using different vaccines together to protect against Covid-19.
This approach, where vaccines work better if a different jab is used for boosting, is known as heterologous boosting and is used in certain types of vaccines such as hepatitis B and tuberculosis.

Boris Johnson: Level of infection is still forbiddingly high
Vaccines minister Nadhim Zahawi said: "This is a hugely important clinical trial that will provide us with more vital evidence on the safety of these vaccines when used in different ways.
"Nothing will be approved for use more widely than the study, or as part of our vaccine deployment programme, until researchers and the regulator are absolutely confident the approach is safe and effective.
"This is another great step forwards for British science, expertise and innovation, backed by Government funding - and I look forward to seeing what it produces."
At Wednesday's news conference, Prof Whitty said the UK was "past the peak" of the current of the pandemic and that cases should continue on a "downward slope" as long as people continued to follow the rules social-distancing.
Mr Johnson, however, has continued to resist calls from some Tory MPs to bring forward the planned re-opening of schools in England from March 8.
The Prime Minister said it was three weeks after the date when the most vulnerable groups - including all over 70s - should have received their first jab, giving time for immunity to kick in.